Recent Advances
PET to ethylene glycol and terephthalic acid
Polyethylene terephthalate (PET) is a polymer most commonly used in beverage and food packaging.
PET can be electrochemically degraded into its original monomers: terephthalic acid (TPA) and ethylene glycol (EG). Luca et al. have developed two methods for this transformation (Scheme 1).
Method one involves the generation of a basic environment through an electrochemical reaction that can subsequently breakdown PET at room temperature (25ºC) without the use of corrosive media.13
Method two depolymerizes PET in high yields via an electron transfer mechanism that is mediated by radical [N-DMBI]. This is an electroorganocatalytic reaction that functions due to the catalytic cycle of [N-DMBI] (Scheme 2).14
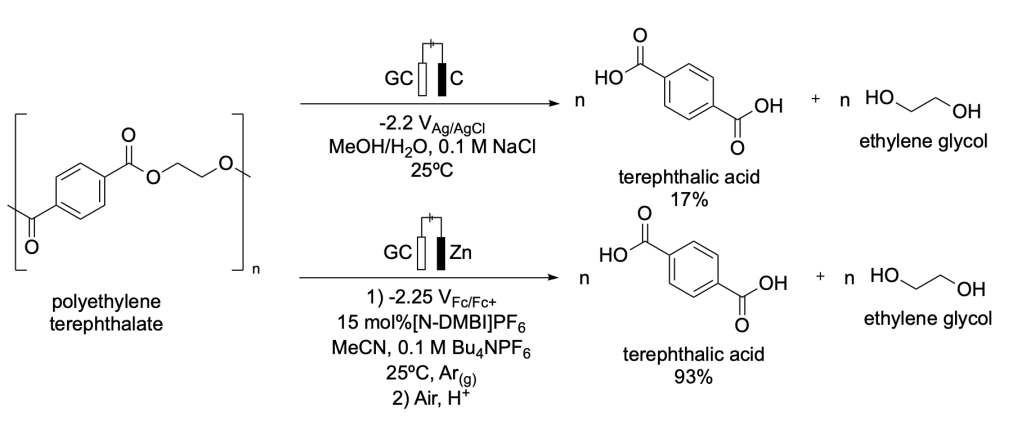
Scheme 1. Reported strategies for the electrochemical degradation of polyethylene terephthalate (PET) to ethylene glycol (EG) and terephthalic acid (TPA).13,14
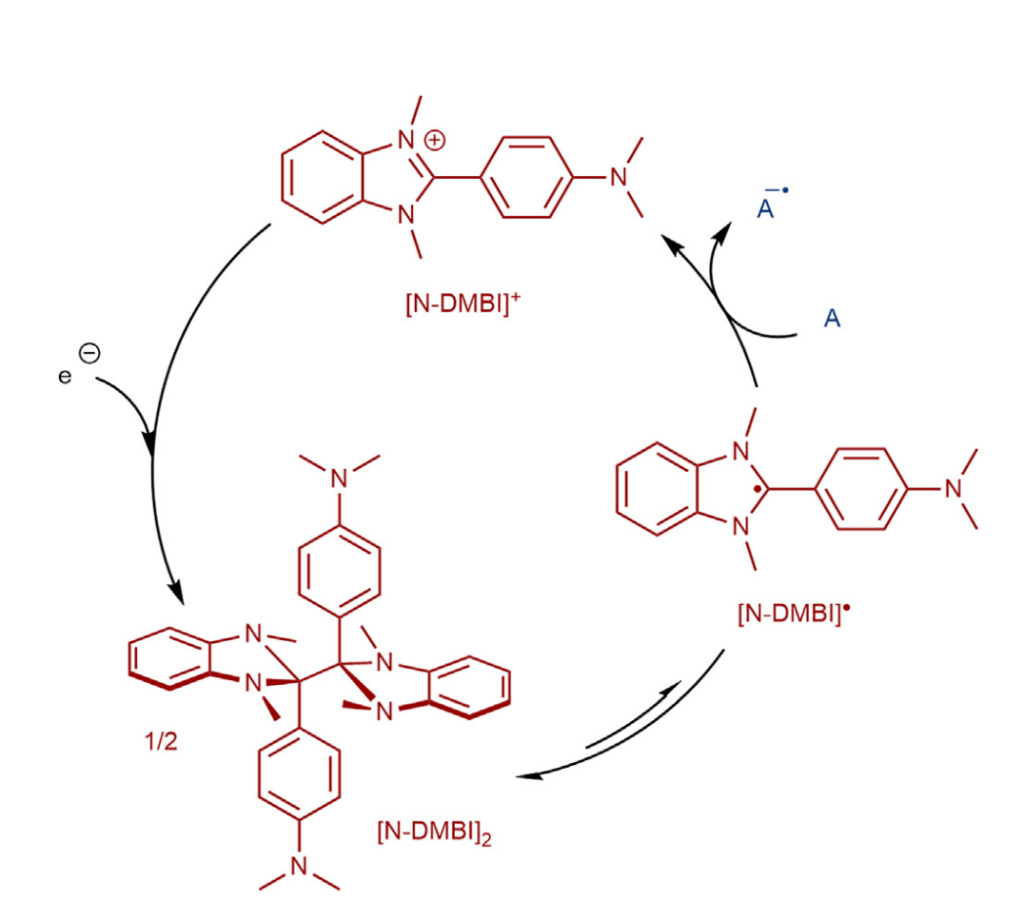
Scheme 2. Proposed catalytic cycle of [N-DMBI] in electrochemical reactions.14
Once PET has been depolymerized, the resulting EG can be further upcycled into high-value products via electrochemical techniques (Scheme 3).
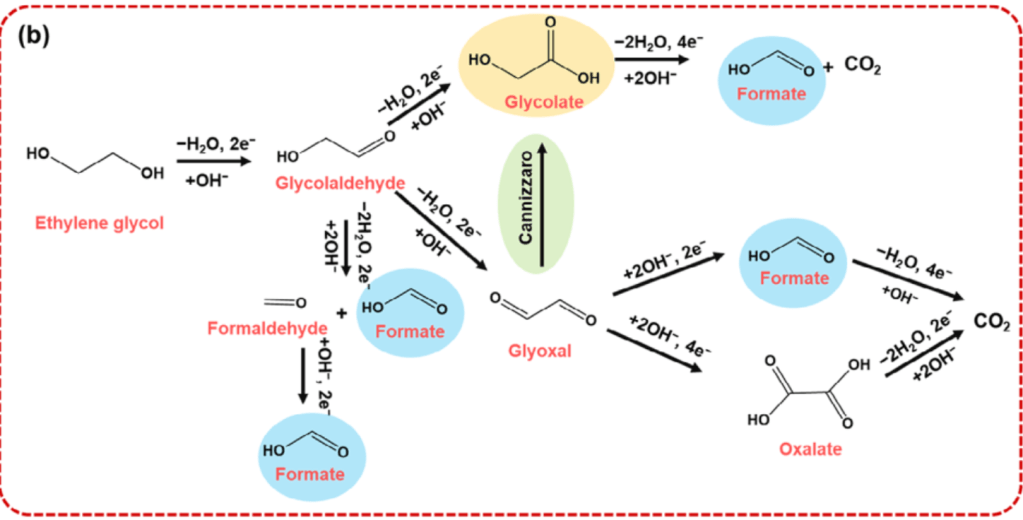
Scheme 3. Electrochemical strategies to convert ethylene glycol (EG) into high-value chemical products.11
POM to formaldehyde, oxydimethanol, and 1,3,5-trioxane
Polyoxymethylene (POM) is one of the most crystalline synthetic polymers. Because of its high crystallinity and resistance to degradation POM is commonly used in mechanical gear.15
Zhou et al. developed a heterogeneous electromediated acid depolymerization mechanism to degrade POM into three high-value chemicals (formaldehyde, oxydimethanol, and 1,3,5-trioxane (Scheme 4).15
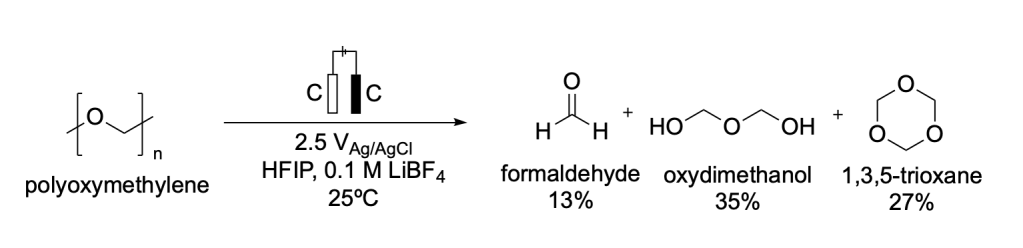
Scheme 4. Reported strategy for the electrochemical degradation of polyoxymethylene (POM) to formaldehyde, oxydimethanol, and 1,3,5-trioxane.
PS to benzoyl products and hydrogen gas
Polystyrene (PS), the polymer found in styrofoam, is widely known as one of the most difficult polymers to recycle, with a recycling rate of 1%. PS also represents 33% of the content in all landfills.17
Hourtoule et al. demonstrated a ferroelectrocatalytic method for the degradation of polystyrene, producing monomeric benzoyl products while simultaneously generating hydrogen gas via a hydrogen evolution reaction (Scheme 5).16
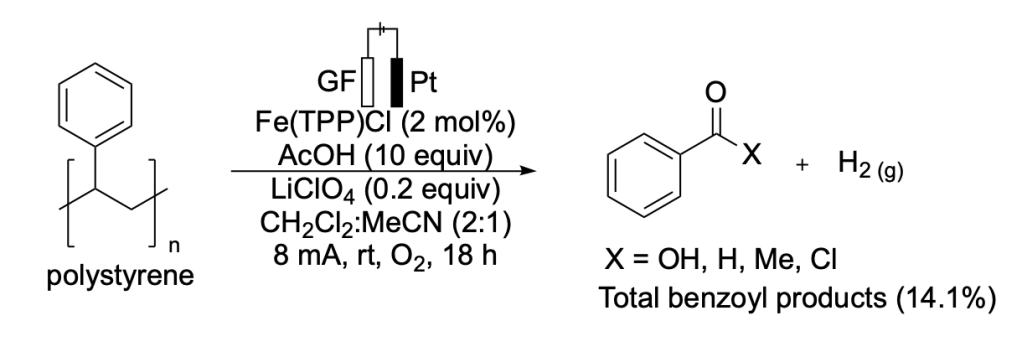
Scheme 5. Reported strategy for the electrochemical degradation of polystyrene (PS) to benzoyl products and hydrogen gas.
This method also proved efficient on a gram scale fueled by solar energy, offering promise for the future of large scale renewable electrochemical recycling.
LDPE upcycling and functionalization
Low-density polyethylene (LDPE) is a thermoplastic composed of polyethylene with branching. LDPE is commonly used in containers and molded lab equipment.
Botte et al. developed an electrochemical oxidation method for low-density polyethylene (LDPE). Rather than completely degrading the polymer, this method introduces ketones, alcohol functionalities, and unsaturation into the polyethylene backbone, using copper electrodes and an acidic environment (Scheme 6).18
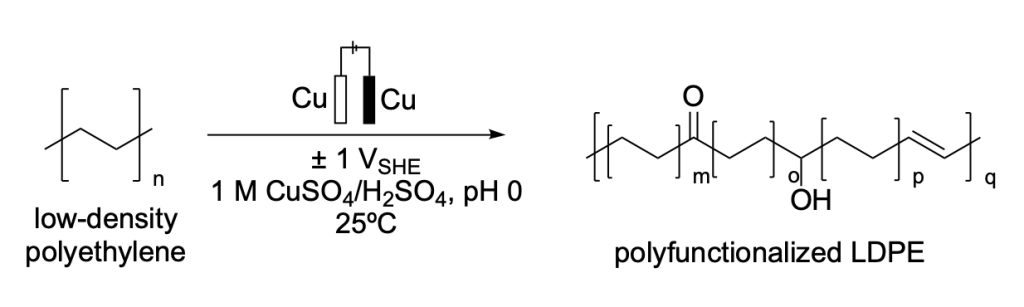
Scheme 6. Reported strategy for the electrochemical upcycling of low-density polyethylene (LDPE) to a polyfunctionalized LDPE.18
This functionalization strategy represents an upcycling approach, where existing plastics can be remodeled into new functional materials.
Possible drawbacks to electrochemical recycling
Although electrochemical recycling shows great promise, there are several challenges that must be addressed before large-scale implementation.
First, energy requirements must be addressed. As in any recycling process energy must be utilized, in this case to supply a voltage to facilitate the transfer of electrons. Fortunately, as proven by Hourtoule et al. solar energy is a promising alternative.
Additionally, electrodes must be manufactured and in some cases may require significant economic input, especially when precious metals such as platinum are necessary.
Finally, large scale electrochemical reactors would need to be built, raising more economic concern.
References
1. Napper, I. E.; Thompson, R. C. Y. 2. Plastics and the Environment. Annual Review of Environment and Resources 2023,48, 55–79.
2. Hutcheon, T. Phase-Out Single use Plastics is the next step in reducing Plastic Pollution. 2019.
https://www.boomerangalliance.org.au/phase-out-single-use-plastics-is-the-next-step-in-reducing-plastic-pollution. (Accessed Feb 18 2026).
3. Zhang, W.; Killian, L.; Thevenon, A. Electrochemical recycling of polymeric materials. Chem. Sci. 2024, 15, 8606–8624.
4. P, G. G.; Bharti, A.; Mondal, A. Electrochemical degradation strategies for polystyrene microplastic: Current trends and future prospects. Polymer degradation and stability 2025, 238, 111351.
5. Yan, Z.; Liu, Y.; Zhang, T.; Zhang, F.; Ren, H.; Zhang, Y. Analysis of Microplastics in Human Feces Reveals a Correlation between Fecal Microplastics and Inflammatory Bowel Disease Status. Environ. Sci. Technol. 2022, 56, 414–421.
6. Geissdoerfer, M.; Savaget, P.; Bocken, N. M. P.; Hultink, E. J. The Circular Economy – A new sustainability paradigm? Journal of cleaner production 2017, 143, 757–768.
7. Wood Mackenzie Chemicals Circular Economy. 2018.
https://www.woodmac.com/news/feature/circular-plastics-economy/. (Accessed Feb 18 2026).
8. Carrette, L.; Friedrich, K. A.; Stimming, U. Fuel Cells: Principles, Types, Fuels, and Applications. ChemPhysChem 2000, 1, 162–193.
9. Frew, J. E.; Hill, H. A. O. Electrochemical biosensors. Anal. Chem. 1987, 59, 933A–944A.
10. Jansson, A.; Möller, K.; Gevert, T. Degradation of post-consumer polypropylene materials exposed to simulated recycling—mechanical properties. Polymer Degradation and Stability 2003, 82, 37–46.
11. Verma, R.; Vinoda, K. S.; Papireddy, M.; Gowda, A. N. S. Toxic Pollutants from Plastic Waste- A Review. Procedia Environmental Sciences 2016, 35, 701–708.
12. Han, S.; Wang, J.; Li, Y.; Wang, C.; Wu, Y.; Liu, B. Strategies for Electrochemical Recycling of Plastic Polyethylene Terephthalate-Derived Ethylene Glycol Into High-Value Chemicals. Adv. Energy Mater. 2025, 15, 2502368.
13. Myren, T. H. T.; Stinson, T. A.; Mast, Z. J.; Huntzinger, C. G.; Luca, O. R. Chemical and Electrochemical Recycling of End-Use Poly(ethylene terephthalate) (PET) Plastics in Batch, Microwave and Electrochemical Reactors. Molecules 2020, 25.
14. Pham, P. H.; Barlow, S.; Marder, S. R.; Luca, O. R. Electricity-driven recycling of ester plastics using one-electron electro-organocatalysis. Chem Catalysis 2023, 3, 100675.
15. Zhou, Y.; Rodríguez-López, J.; Moore, J. S. Heterogenous electromediated depolymerization of highly crystalline polyoxymethylene. Nature Communications 2023, 14, 4847.
16. Hourtoule, M.; Trienes, S.; Ackermann, L. Anodic Commodity Polymer Recycling: The Merger of Iron‐Electrocatalysis with Scalable Hydrogen Evolution Reaction. Angewandte Chemie International Edition 2024, 63, e202412689–n/a.
17. Rahimi, A.; García, J. M. Chemical recycling of waste plastics for new materials production. Nature Reviews Chemistry 2017, 1, 0046.
18. Rahimzadeh, R.; Ortega-Ramos, J.; Haque, Z.; Botte, G. G. Electrochemical Oxidation and Functionalization of Low-Density Polyethylene. ChemElectroChem 2023, 10, e202300021.